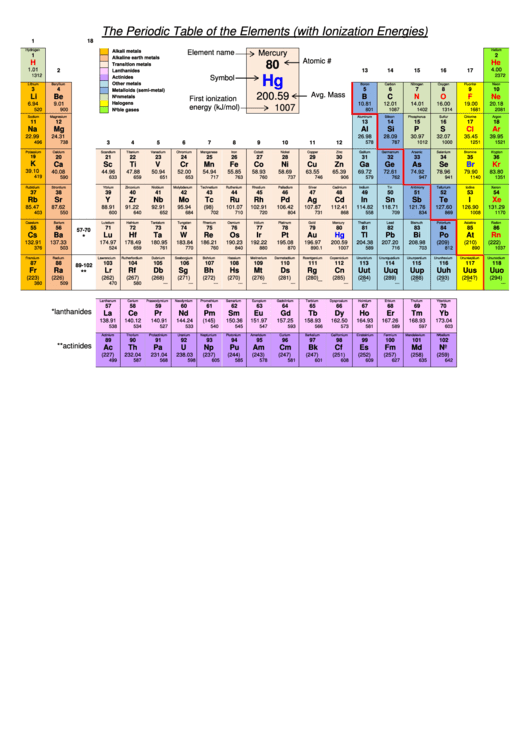
After the first ionization energy it becomes significantly harder for electrons to be removed because taking away electrons will result in closer attraction between the nucleus and the electrons as well as a smaller shielding effect because there is not enough electrons to block attraction between one another. After the first ionization energy, the trend continues but in a less pronounced way when used in the second or third ionization energy. There are more electrons the farther down the periodic table thus a larger shielding effect occurs causing for less ionization energy to remove an electron. The shielding effect is the blockage of of electrons attraction of those in the outer energy level with the nucleus. X + energy X + + e where X is any atom or molecule capable of being ionized, X + is that atom or molecule with an electron removed (positive ion), and e is the removed electron. The second reason that the ionization energy decreases down a group is the shielding effect. Periodic Table Ionization Energy Ionization energy, also called ionization potential, is the energy necessary to remove an electron from the neutral atom. The lack of energy levels causes greater attraction between the nucleus and the electrons causing the need for more ionization energy to remove and electron. There are less electrons thus there is no need for more energy levels. The atomic radius is smaller at the top of the periodic table. The reason for this is shielding and atomic radius. The trend for ionization energy in groups is the the ionization energy decreases from from top to bottom, meaning that the highest ionization energy is at the top and the lowest ionization energy is at the bottom. This greater pull or attraction requires more energy to remove an electron from an atom. The atomic radius hold more nuclear charge or more protons thus causing a greater attraction or pull between the protons and the electrons. When the nuclear charge is greater, the atomic radius is smaller. This is not two reasons because the two coincide with each other. This reason being the atomic radius and the nuclear charge. The ionization trend for periods is from left to right, the ionization energy increases.The ionization energy increases for one main reason. The third ionization energy is the energy required to remove an electron from an Aᐩ². Ionization energy is measured in kJ/mol The second ionization energy is the energy required to remove an electron from an Aᐩ¹. The shielding affect makes it easier to remove the outer most electrons. Shielding Effect is when electron and nucleus in an atom have an decrease in attraction. The reason as to why Ionization Energy decreases going down the periodic table is because of shielding effect. This is known as the first ionization energy. Ionization Energy increases as you left to right of the periodic table. Ionization energy is the energy required to remove one electron from a neutral atoms of an element.
